Our Technology
Re-Vana’s proprietary technologies are based on a novel photocrosslinked technology.
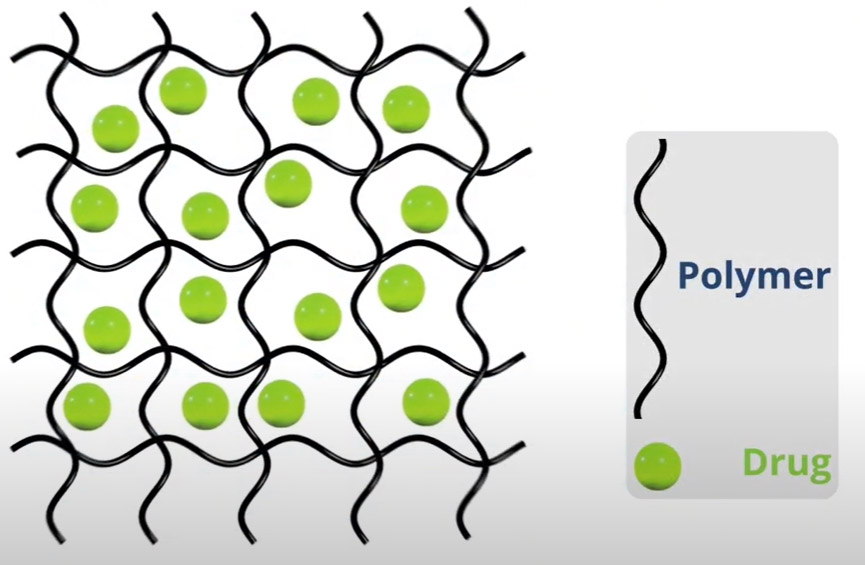
Our unique photocrosslinked implants enable us to manufacture products at room temperature and with virtually no change in internal pH. Through photocrosslinking the implant restricts water movement, thereby maintaining stability of the active drug within the implant matrix and controls the degree of swelling. This results in enhanced protein stability for extended drug delivery. The degree of photocrosslinking, percentage of drug loaded into the implant, and implant dimension enables Re-Vana to customize the rate and duration of drug delivery.
This novel approach to sustained drug delivery can be used for a wide spectrum of large and small molecule therapeutics. Once injected into the eye, the implant slowly biodegrades, releasing the drug at controlled rates for extended periods of time.
New Re-Vana technology animation demonstrates EyeLief® and OcuLief® delivering sustained release biologics.
Re-Vana’s sustained release products can be delivered into the eye in a pre-formed implant or via an in situ depot forming implant that is photocrosslinked during injection into the eye. This enables Re-Vana to deliver larger volumes of drug for extended therapeutic benefit.
